Graphene is an amazing conductor. The transport of electrons through graphene nanoribbons has even surpassed what scientists thought were the theoretical limits for the material—so much so that electrons moving through it seem to behave almost like photons.
Graphene’s amazing properties as a conductor has inspired some researchers to explore whether the single-atom-thick sheets of carbon could also be made into superconductors. Last year, an international research team from Canada and Germany was able to demonstrate that graphene can be made to behave that way when it’s doped with lithium atoms.
Now researchers in Japan (from Tohoku University and the University of Tokyo) have developed a new method for coaxing graphene to behave as a superconductor that has some important and distinctive differences from the previous research by the Canadian and German researchers.
“The most decisive difference is that we have observed zero electrical resistivity while the Canada and German team has not,” said Takashi Takahashi, a professor at Tohoku University and leader of the research, in an e-mail interview with IEEE Spectrum. Asked what’s behind that claim, Takahashi said:
[The Canadian-German research team] observed only an energy gap by ARPES at low temperature that may be an indirect signature for superconductivity. But, an energy gap opens at low temperature even when another different ordering such as a charge-density-wave emerges, [turning the sample into] a semiconductor, not a superconductor. They were not able to exclude this possibility.
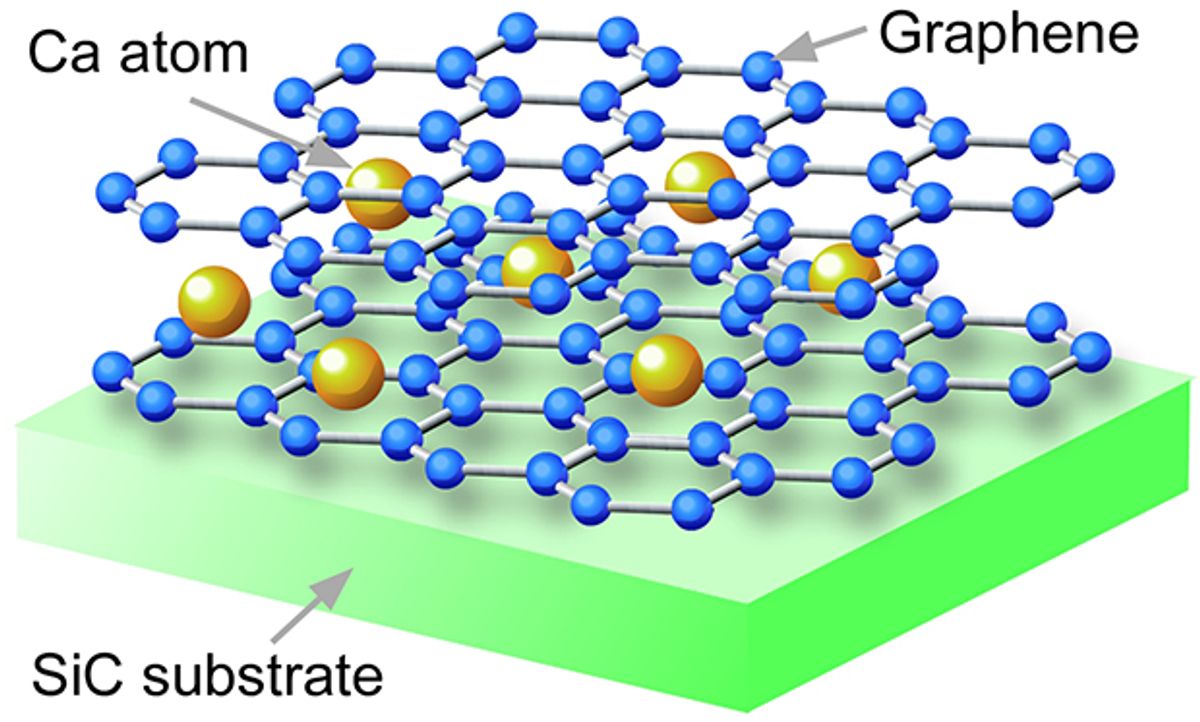
To ensure the to ensure that they had an actual superconductor, Takahashi’s team, in research described in ACS Nano, took a number of different approaches to preparing their material. First, they fabricated high-quality graphene on a silicon carbide crystal, and controlled the number of graphene sheets. This gave them a well-characterized bilayer graphene, into which they stuffed calcium atoms. So careful were Takahashi and his team in formulating the crystal structure of the graphene-calcium sandwich and counting the number of calcium atoms, that they could actually ascribe a chemical formula to their sample: C6CaC6.
“This is very important because the amount of Li or Ca atoms determines the amount of donated electrons into graphene, which controls the occurrence of superconductivity,” said Takahashi.
Measurements confirmed that superconductivity did occur with this latest formulation of graphene. The researchers discovered that the electrical resistivity dropped rapidly at around 4 K (-269 °C), indicative of an emergence of superconductivity. The measurements further indicated that the bilayer graphene did not create the superconductivity, nor did lithium-intercalated bilayer graphene exhibit superconductivity. This meant that the drop in resistance was due to the electron transfer from the calcium atoms to the graphene sheets.
Now that graphene has been made to perform as a superconductor with a clear zero electrical resistivity, ideas about potentially applying into making a quantum computer that would use this superconducting graphene as the basis for an integrated circuit.
Of course, the temperature at which the graphene transitions into a superconductor is still very low to be practical for any such device and this issue will be the next point of interest for the researchers.
Takahashi added: “We will look to increase the superconducting transition temperature, for example, by using different metals and/or changing the number and stacking sequence of the graphene sheets.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.