Good-bye, Wheelchair, Hello Exoskeleton
This year, the Ekso Bionics exoskeleton for paraplegics hits the market

In a warehouse that looks like a cross between a mad inventor’s garage and a climbing gym, a pair of mechanical legs hangs from the ceiling on ropes. With the quiet whir of four motors, one in each hip and knee, the legs take a step, then another and another. This is an exoskeleton walking suit, and it is taking the hundreds of thousands of steps that regulators demand to prove that it’s no mere toy but a reliable medical device, one that just might change the lives of people who thought they’d never again rise from a wheelchair.
The Berkeley, Calif., warehouse is the home of Ekso Bionics (formerly known as Berkeley Bionics), a young company that’s about to step out onto the world stage. Early this year the company will begin selling its Ekso suit to rehab clinics in the United States and Europe, to allow patients with spinal cord injuries to train with the device under a doctor’s supervision. By the middle of 2012, the company plans to have a model for at-home physical therapy.
When you don the Ekso, you are essentially strapping yourself to a sophisticated robot. It supports its own 20-kilogram weight via the skeletal legs and footrests and takes care of the calculations needed for each step. Your job is to balance your upper body, shifting your weight as you plant a walking stick on the right; your physical therapist will then use a remote control to signal the left leg to step forward. In a later model the walking sticks will have motion sensors that communicate with the legs, allowing the user to take complete control.
“We took the idea of the external skeleton, and we added nerves in the form of sensors and motors that represent your muscles and computers that represent your brain,” says Eythor Bender, CEO of Ekso Bionics.
The company began its evolution in 2005 with the ExoHiker, an exoskeleton that allows able-bodied people to carry 90 kg (about 200 pounds) with minimal exertion. The company’s engineers at first thought it would take 5 kilowatts to power such an exoskeleton, which would have meant bulky batteries and motors. The breakthrough was a redistribution of weight that reduced the power requirements by three orders of magnitude. A later system, the load-carrying HULC (Human Universal Load Carrier), was licensed to Lockheed Martin Corp. for military development in 2009, and Ekso Bionics’ engineers began looking for a new direction. Their energy-efficient devices, they realized, left them with a “power budget” that could be spent on moving the user’s legs. That’s when paraplegic people became the company’s target customers.
A few other companies around the world are bringing out exoskeletons for people with disabilities, but Ekso Bionics’ push in 2012 may give it a market advantage. Ten top U.S. rehab clinics have already signed up for the first batch of production units.
One of the first devices will go to Mount Sinai Hospital, in New York City, where Kristjan T. Ragnarsson, chairman of the department of rehabilitation medicine, has treated spinal cord patients for 40 years. His patients’ priorities have never changed. “The first thing they want to know is whether they will walk again,” says Ragnarsson. “As their physician, I always have to address that question.”
Over the years he has told his patients about the latest inventions, from stiff air-filled garments to devices that electrically stimulate the muscles, but all these contraptions proved too difficult for the patients to operate. “They were completely exhausted after just a few steps,” he says.
Ragnarsson thinks the Ekso can succeed where so many others failed, because the powered device does most of the labor for the patient. “I’m optimistic, actually, that this will work,” he says. “I think my patients will be able to stand up and take a few steps and face the next person directly on!”
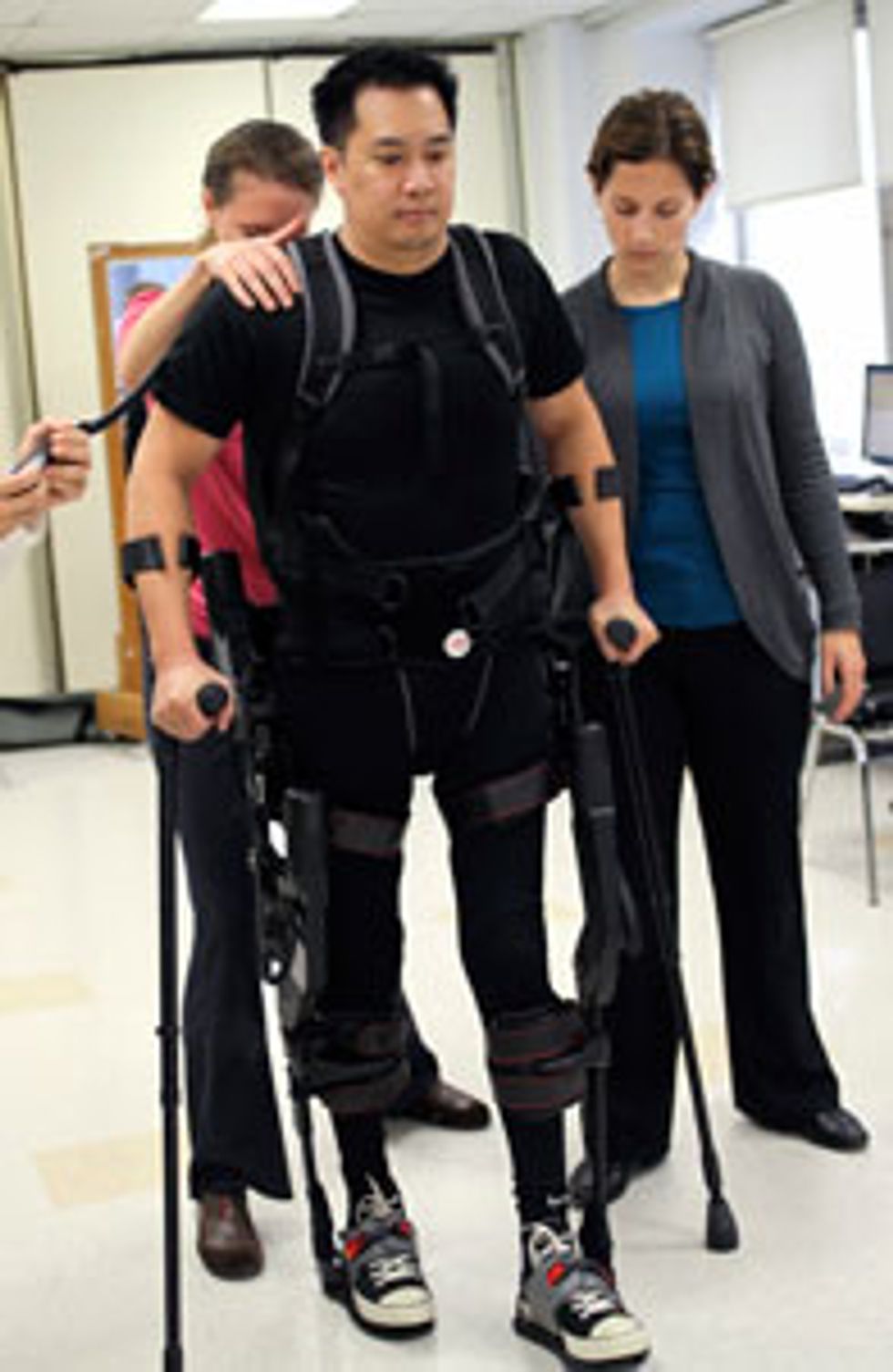
In a rehab room at Mount Sinai Hospital in November, Robert Woo, an architect, is justifying his doctor’s optimism. His high-tech wheelchair, which his kids have plastered with stickers, sits in the corner. That wheelchair has been his only source of mobility in the four years since a construction crane dropped 6 metric tons of metal on his job-site trailer, crushing his spine.
But today Woo stands tall. He leans forward on the two walking sticks as physical therapists and Ekso technicians buzz around him. Standing in the Ekso feels like floating, Woo explains, and he’s still getting used to it. “I’m always looking down, wondering, How are my feet doing?” he says with a smile. “I have to build up confidence in myself and confidence in the device.” Then a therapist calls out, “Step!” and punches a button on her controller. With the creak of plastic joints and the whir of motors, Woo is walking.
His progress is slow and jerky, and on one step he leans too far forward and starts to lose his balance before the therapists grab his arms and steady him. But most of the 308 steps Woo takes during the hour-long session are remarkably fluid, with a heel-to-toe gait. Achieving that natural stride was a major technological challenge, as it requires a sophisticated transition of control among the four servomotors that supply the torque to the knee and hip joints.
The session was only Woo’s third time using the device, but he had already graduated from a wheeled walker to the walking sticks. He’ll have to wait a bit before he makes any more progress: The Ekso was at Mount Sinai for only a week’s tryout. Ragnarsson expects the hospital to buy a device of its own this year for the steep price of about US $100 000; that price should fall as production increases.
The company expects to test its physical therapy model soon on patients with other diagnoses, like multiple sclerosis and stroke. And by 2014, it plans to release a personal model that can be used not just for rehab but for everyday living. Woo says he looks forward to a day when he can stand in the Ekso and cook dinner in his kitchen or take a walk in the park with his children.
Before that day comes, though, Ekso Bionics must get approval from the U.S. Food and Drug Administration by proving both the device’s safety and its benefits. And its engineers must work out how the user will initiate more complicated movements, like climbing stairs and sitting down.
Bender, Ekso Bionics’ CEO, is confident that controlling Eksos will come to feel utterly natural for the people who rely on them for mobility. “People talk about robots taking over, especially in factories, and taking away our jobs. But the way we see it is, if you can’t beat them, join them!” says Bender. “We are joining with the robots. And this has the ability to make us stronger, more productive, and to improve our quality of life.”
This article originally appeared in print as “Good-bye, Wheelchair.”