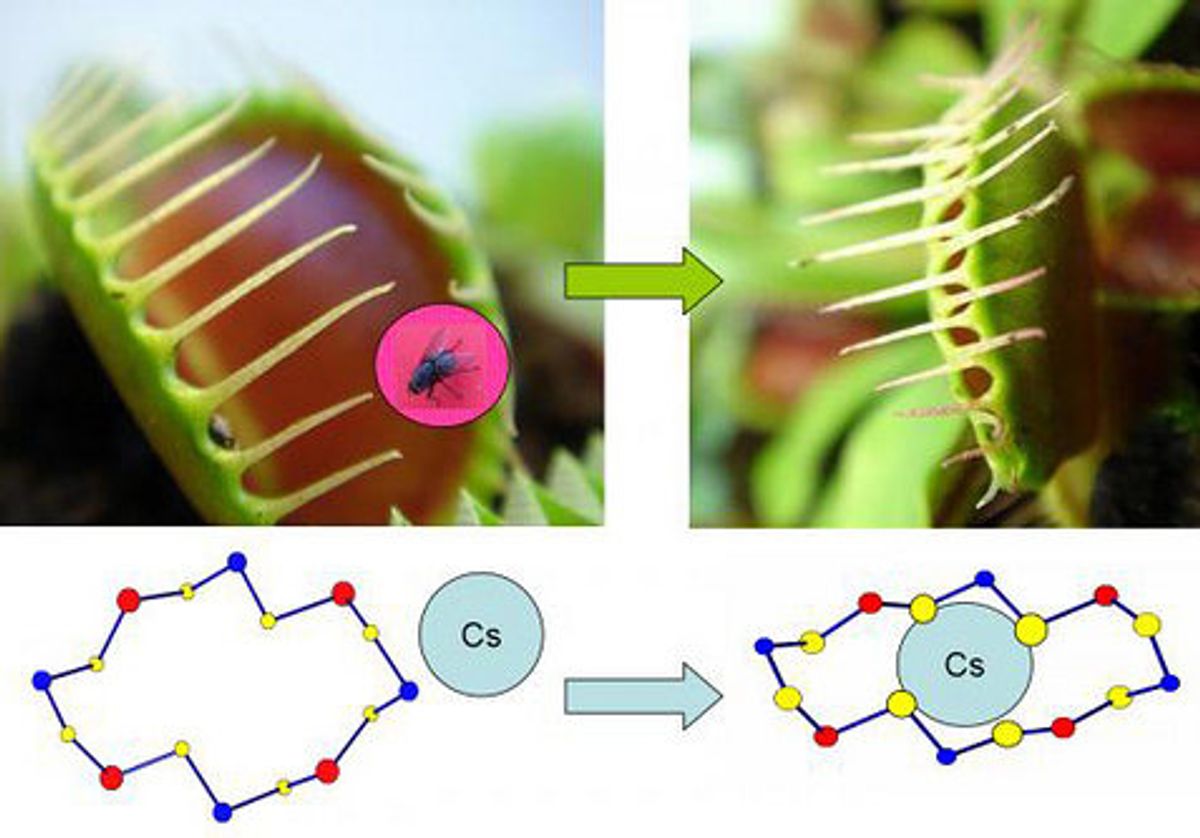
Researchers at the Argonne National Laboratory near Chicago created a rigid structure that carries a negative charge. The substance, made of layers of [(CH3)2NH2]+ and [Ga2Sb2S7]2−, has pores which attract positively charged ions when in solution. Sodium ions, which account for the harmless part of nuclear waste, move freely in and out of the framework when it is immersed in water. The other part, though, is not harmless: cesium atoms are highly radioactive, can leach into soil and water and stay in the environment for decades (like they have at Chernobyl). They also bonded to the sulfide framework, but they didn't escape like the sodium could.
"Imagine the framework like a Venus flytrap," said one of the researchers involved, Mercouri Kanatzidis, in a press release. "When the plant jaws are open, you can drop a pebble in and the plant won't close—it knows it isn't food. When a fly enters, however, the plant's jaws snap shut." The research was published in Nature Chemistry.
Cesium does not bond as freely with water as does sodium, which explains the difference when the cesium atoms enter the framework and can't get out again. The authors say this could eventually be used to aid in cleanup of nuclear waste or disaster zones.
"The dynamic response we describe here provides important insights for designing new materials for the selective removal of difficult-to-capture ions," the investigators wrote in the paper's abstract.
Photo via Argonne Nat. Lab on Flickr.
Dave Levitan is the science writer for FactCheck.org, where he investigates the false and misleading claims about science that U.S. politicians occasionally make.