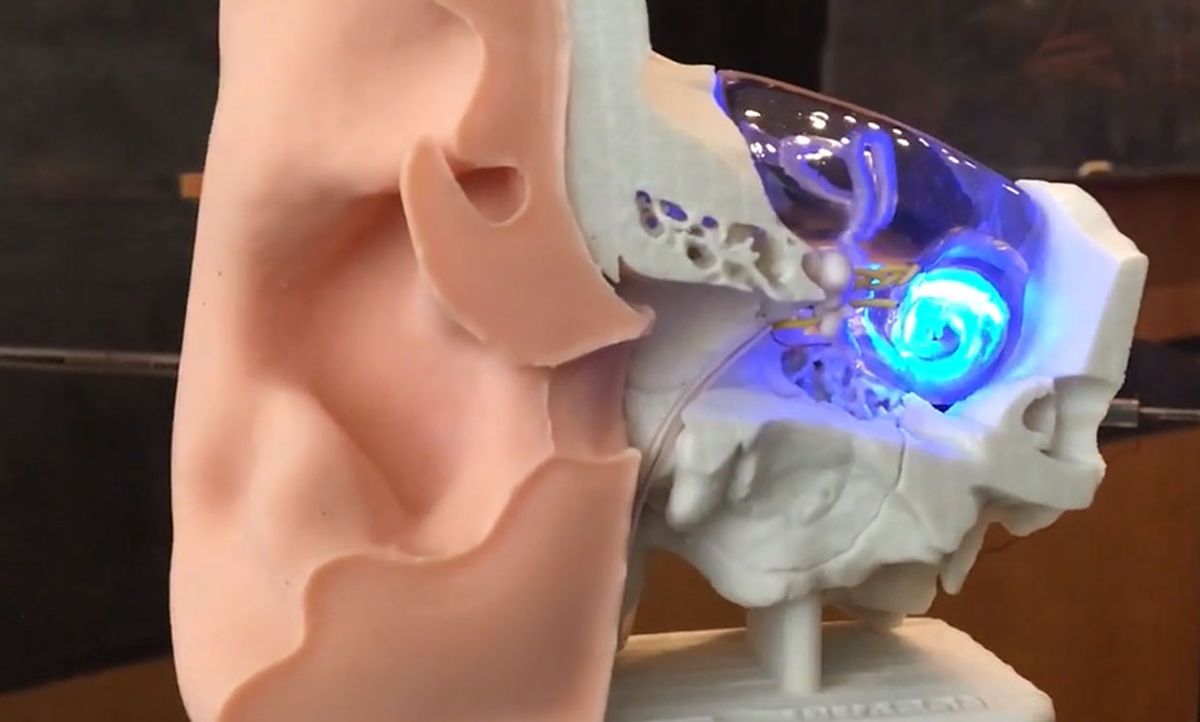
Scientists in Germany have succeeded in restoring hearing sensations in gerbils using flashes of light. The technique, if it can be developed for humans, could offer a more refined, high-resolution auditory experience than what can be achieved with current hearing devices such as the cochlear implant.
The scientists, led by Tobias Moser, a professor of auditory neuroscience at University Medical Center Göttingen, achieved the effect using optogenetics. The technique involves genetically altering specific neurons so that they respond to light and has become one of biotech’s hottest tools.

In this case, the researchers altered auditory neurons, and then controlled them with light delivered with implanted optical fibers. They described their experiments today in the journal Science Translational Medicine.
Moser and his colleagues are attempting to solve a problem that has stumped engineers for decades. The cochlear implant, invented half a century ago, does a decent job of sensing and converting speech in an otherwise quiet space. But move that conversation into a crowded room and the wearer hears a jumbled mess.
“The chief complaint of people who depend on cochlear implants is that it’s hard to understand speech in noise,” says Moser. That’s because cochlear implants stimulate the ear too broadly. The device works by converting sound to electrical impulses that are delivered to the inner ear with electrodes. These impulses stimulate auditory nerves that send messages to the brain, creating the experience of sound.
The problem is that the signals generated tend to spread widely around each electrode contact in the ear. The signals then interfere with one another, limiting the number of electrodes that can be used at a given time. As a result, the signals lump together many sound frequencies, making it difficult to discern between similar sounds due to poor frequency resolution. (You can experience a simulation of the sound from a cochlear implant by downloading this software.)
Researchers have tried to improve the device with better stimulation techniques, including multipolar stimulation and electrodes that penetrate the auditory nerve. Those advances have improved the device, but none has solved the background noise problem.
“Optical stimulation may be the breakthrough to increase frequency resolution,” Moser says. The technique, called optogenetics, can be applied to cells in countless ways. It has been used, most notably, to hack into animal brains to manipulate memories, behavior, and pain.
Moser thinks the technique can manipulate the auditory pathway too. By genetically engineering certain auditory neuron populations to respond to light, the researchers can selectively control the activation of those neurons. This should enable them to generate more frequency information that the brain can better discriminate.

He and his colleagues chose to demonstrate the idea in gerbils. To engineer the animals’ auditory cells, the researchers injected into the cochlea of their ears a virus that carries genetic coding for light sensitivity. With that coding, the cells become sensitive to light. Then the researchers implanted optical fibers in the ear to deliver light to those neurons.
When they turn the light on, the neurons fire action potentials that travel to the brain, creating the experience of sound. The gerbils don’t see the light, but they hear it.
To test the system, the scientists trained the gerbils to jump to another compartment of their enclosure after optical stimulation of the ear. Then they played an audible alarm. The animals still jumped, demonstrating that the optical device indeed sent soundlike signals to the gerbils’ brains. A similar experiment worked in deaf gerbils.
It’s unclear exactly what kind of sound the gerbils perceived from the optical stimulation—parsing that out is something for future studies. Moser says at this point he’s just trying to show that the technique works at all.
Previously he and his colleagues had demonstrated the concept in genetically engineered mice and rats. Today’s experiments showcase the tool in a better animal model, with improved stimulation timing.
The experiments prove that the technology is worth pursuing in larger animal models, Moser says. Next, he and his collaborators plan to increase the complexity of the signals and apply the technology to marmoset monkeys.
There’s a bit of a competition going on in Moser’s field. Another group, led by Claus-Peter Richter, an auditory researcher at Northwestern University in Chicago, is also pursuing light as an alternative to the electrical cochlear implant. But instead of optogenetics, Richter’s group is using near-infrared lasers.
In that technique, the infrared light is absorbed by the liquid inside the cochlea, generating heat. In the cochlea of deaf animals missing their hair cells, the heat changes the membrane properties of the auditory neurons, generating an electrical signal that communicates with the brain, according to Richter.
Richter says Moser’s experiments are “a great step forward” in the optogenetic technique. But he says he prefers the near-infrared method because it doesn’t involve injecting a viral vector in the body—something that is technically difficult and hasn’t been proven in human studies.
The near-infrared route, which Richter pioneered, faces its own challenges. Namely, the energy requirements for powering the laser may be impractically high, says Moser. And while Richter says he has been able to get the near-infrared technique to work in auditory nerves of deaf animals, three other groups have not been able to repeat it. The effect generated in the animals may actually be a kind of acoustic effect rather than a direct optical effect, some researchers say.
“I essentially took the harder route with optogenetics because it has a mechanism I understand and the power requirements are more reasonable,” says Moser.
The discussion will surely continue to play out in the literature. “Each method has its benefits and challenges,” says Richter. “I’m keeping my eyes open to see what the competition is doing.”
Emily Waltz is a features editor at Spectrum covering power and energy. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.