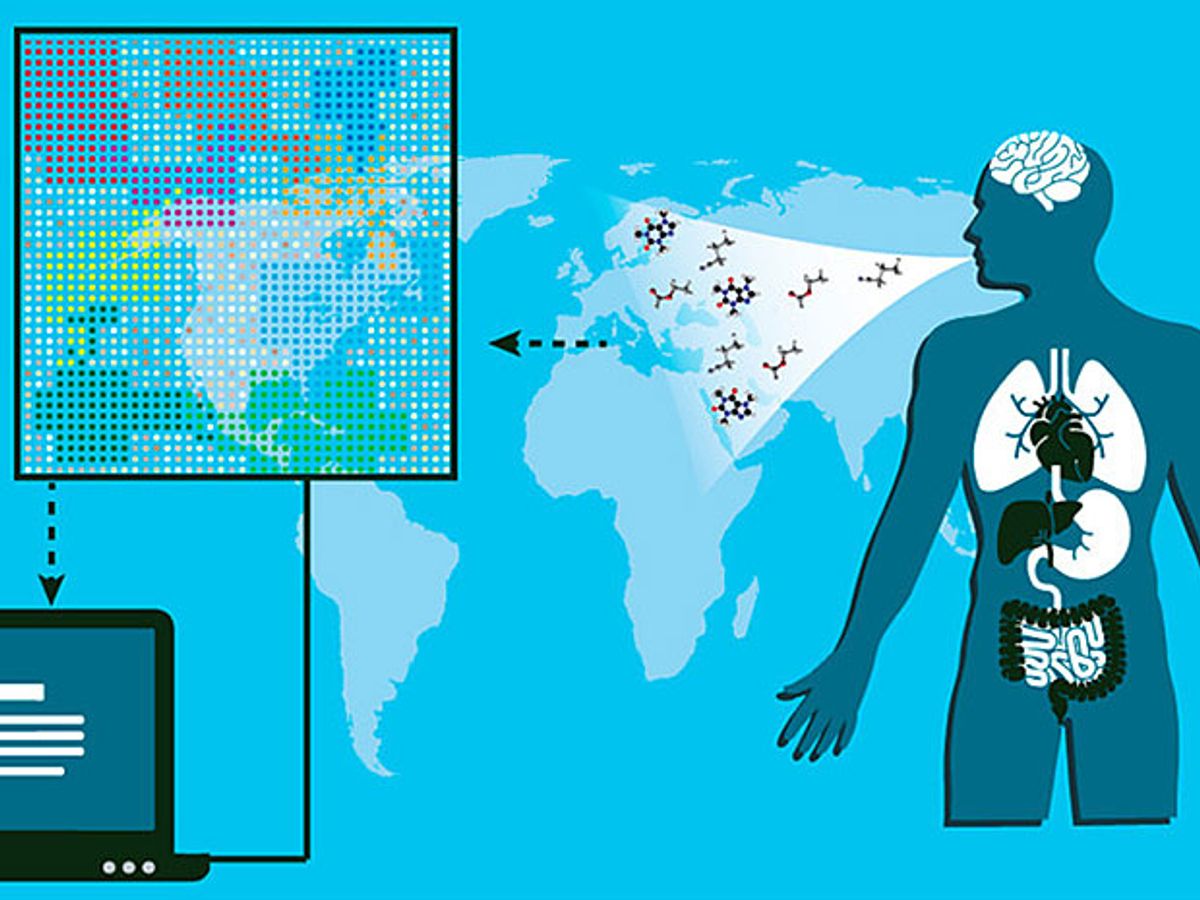
Imagine talking to your best friend on the phone, when suddenly a text message from your doctor pops up. A plug-in module on your smartphone has detected an unusual chemical pattern in your breath, and you need to come in to be evaluated for early signs of cancer.
Physicians have been detecting disease from the smell of breath, urine, and feces for over 2,000 years. Illness can change the metabolism in our bodies, causing our cells to release volatile organic compounds (VOCs), molecules that travel through the bloodstream to the lungs and are exhaled. People with early stage cancer, for example, breathe out different concentrations of VOCs than healthy individuals.
In dozens of studies, scientists have detected VOCs in the breath of patients using laboratory techniques like gas chromatography and mass spectroscopy. Now, a team of researchers, led by engineer Hossam Haick of the Technion−Israel Institute of Technology in Haifa, reports its latest advances with a simpler, more sophisticated method for detecting disease in the breath. Called the “Na-Nose,” this artificially intelligent nanoarray relies on gold nanoparticles and carbon nanotubes to diagnose and classify 17 different diseases based on a single human breath.
In a December study published in ACS Nano, the researchers for the first time demonstrate clinical proof that diseases have unique chemical patterns, or “breathprints,” that distinguish them from one another. In the study, the Na-Nose used those patterns to distinguish among diseases with 86 percent accuracy, and showed potential to diagnose more than one disease at once.

Haick began developing the device 10 years ago when he joined the faculty at Technion after completing a postdoc at the California Institute of Technology. The screening tool is made up of two parts: a white, desktop box with tube into which a person exhales, sending his or her breath into an array of sensors; and an attached computer with machine-learning software trained to recognize patterns from those sensors.
The array consists of thin layers of either gold nanoparticles or carbon nanotubes, each coated with organic ligands—sticky molecules that bind compounds in our breath. When VOCs in the breath bind to the ligands, it changes the electrical resistance between the nanoparticles or nanotubes, and that signal is sent to a computer. There, pattern-recognition software determines if the signal corresponds with a known chemical signature of a particular disease.
The lab has trained the device on over 23 illnesses, teaching it to discriminate between a healthy individual and an individual with one of these catalogued diseases. But “that’s the easy part,” says Haick. Next, his team took the device into clinics, testing on over 8,000 patients to teach the software to discriminate between disease and confounding factors, such as contamination, age, gender, background disease (such as obesity or diabetes) and geography. And it worked: Last year, for example, the team found that the tool could detect gastric cancer in a blinded test of patients with 92- to 94-percent accuracy.
The current study, for the first time, used the Na-Nose to detect and discriminate among 17 different diseases in the breath of 1,404 individuals across five countries. “For every disease, there is a unique fingerprint throughout exhaled breath that is quite distinguishable from other disease types,” says Haick.
That’s not to say physicians will soon be able to diagnose everything through breath. It has been difficult to accurately diagnose prostate, breast, and bladder cancer based on exhalations, says Haick, so it is likely that other tests, such as urine analysis, may be better suited in those cases.
He is now working to miniaturize the device in the hopes of adding a module onto smartphones. The project, called SniffPhone and funded by the European Union’s Horizon 2020 Program, is currently underway, but Haick expects the desktop box will reach doctor’s offices much sooner.
When it does, he hopes the Na-Nose will help doctors catch signs of cancer in people long before other types of tests would be able to. In clinical studies, it was able to discriminate among stages of gastric cancer and lung cancer as early as stage one. “We aim to catch disease at an early stage, where we can increase the survival rate,” says Haick.
Megan is an award-winning freelance journalist based in Boston, Massachusetts, specializing in the life sciences and biotechnology. She was previously a health columnist for the Boston Globe and has contributed to Newsweek, Scientific American, and Nature, among others. She is the co-author of a college biology textbook, “Biology Now,” published by W.W. Norton. Megan received an M.S. from the Graduate Program in Science Writing at the Massachusetts Institute of Technology, a B.A. at Boston College, and worked as an educator at the Museum of Science, Boston.