At the end of Star Wars Episode V: The Empire Strikes Back, Luke Skywalker feels when a needle pricks his newly-installed bionic hand. Researchers report today in the journal Science Translational Medicine that they can do something similar: stimulating regions of a human test subject’s brain with electrodes can recreate the perception of touch in a robotic hand.
This year, about 280,000 people in the United States alone are living with a spinal cord injury, according to the National Spinal Cord Injury Statistical Center. Depending on the severity, damaged nerve connections lead to effects ranging from a partial loss of feeling to complete loss of motion in different limbs.
“If you lose that sense of touch, you have a really difficult time” grabbing, holding, and manipulating different objects, says Richard Gaunt, a neuroengineer at the University of Pittsburgh who works on touch feedback for prosthetics. To safely interact with other human beings and handle delicate objects such as eggs, humans need more than just control–they need to be able to modify control based on touch feedback.
In the May 2016 issue of IEEE Spectrum Dustin Tyler, head of the Functional Neural Interface Lab at Case Western Reserve University in Cleveland, described technology that allows amputees to feel a robotic arm prosthesis. That technique relied on electrically stimulating peripheral nerves using electrodes implanted into the user’s arm. The system improved the delicate motion capabilities in a robotic arm. But the technology doesn’t work if the connection between limbs and brain is absent, such as with spinal cord injuries, Gaunt says.
“We’re going straight to the brain,” he says.
In the early 20th century, surgeons first began stimulating the cortex of the human brain to help treat epilepsy and understand motor function. Research published in Nature in 1997 suggested that animals exposed to electrical stimulation in specific regions of the brain react as though their hands were stimulated, but even with follow-up studies, exactly what animals feel remained a mystery. In several experiments where scientists electrically stimulated human brains, such as 2013 research in the Journal of Neural Engineering, participants reported sensations that felt like they were coming from the hands, but described the feelings a more of a buzzing or the tingling sensation, like when your foot falls asleep.
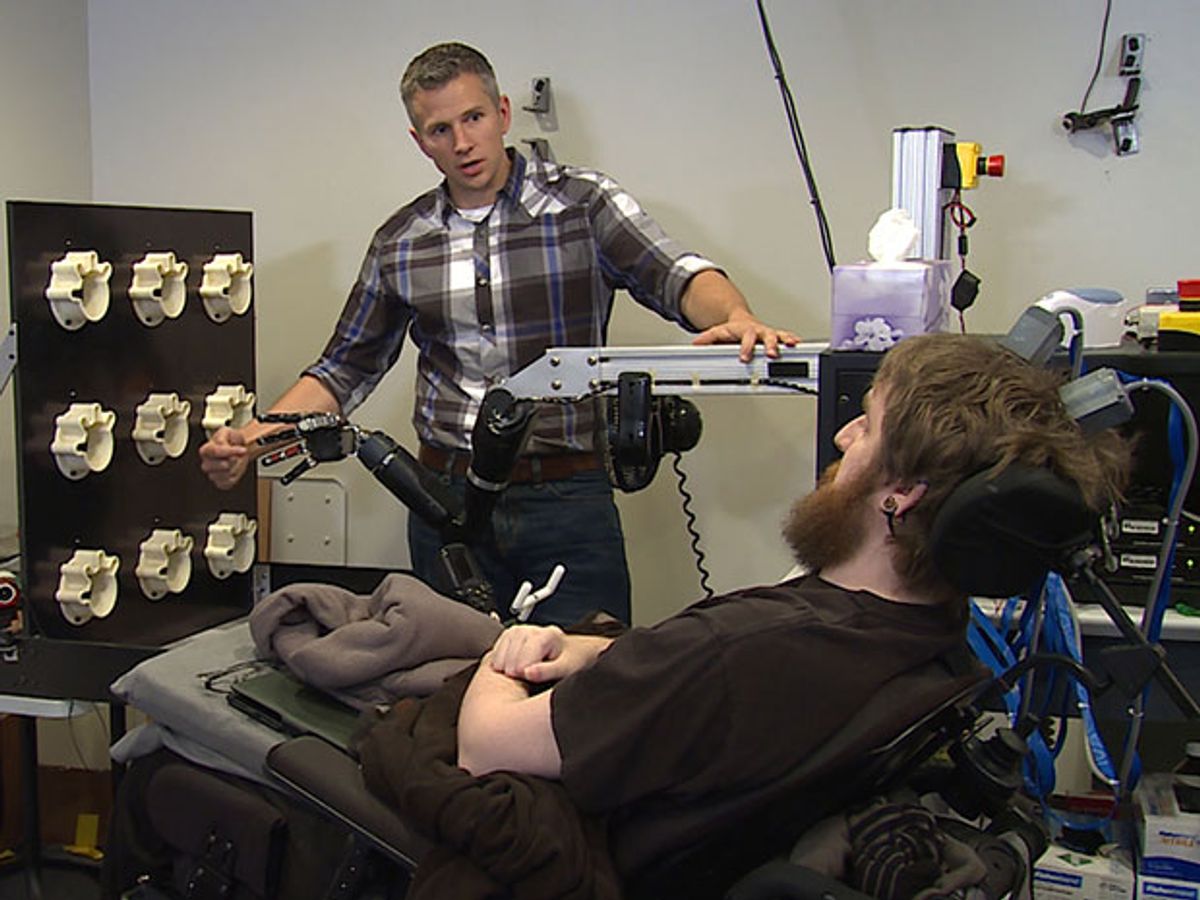
In the new research, Gaunt and his team ran a six-month experiment on a man who suffered a spinal cord injury about 10 years prior. They created a map linking brain regions and finger sensations, which they used to simulate touch in a robotic hand.
The core of the system consisted of two neural implants: 2.4 mm by 4 mm microelectrode arrays with 60 electrodes. But where to put them?

The researchers began the experiment by tracking the magnetic fields coming from the brain’s neurons when the test subject, Nathan Copeland, imagined something touching different parts of his hand. By combining the readings with magnetic resonance imaging, they created a map of groups of neurons in the S1 cortex associated with feeling in the thumb, index finger, little finger, and palm.
Guided by the map, the researchers cut into the test subject’s brain, inserting the implants about a sheet of paper away from the relevant neurons. The microelectrode arrays linked up to metal pedestals that stuck outside his skin for connection to an external electric stimulator.
For the first couple of weeks after the surgery, Copeland described tingling throughout the entire body, then after only in the hands. This was caused by random neurons firing, which eventually died down.
The team connected Copeland’s implants via cables to a micro-stimulation system. They began stimulating neurons with little pulses of electricity and tracked the subject’s responses.
Over the next few weeks, during stimulation the patient reported variations of pressure, touches, vibrations, and tingles that felt as if they occurred at joints and below the skin of a hand. The mappings between brain regions and hand sensations were fairly consistent over the six months, Gaunt says.
For the grand test, they blindfolded the subject and hooked him up to a robotic hand. When they pressed one of the fingers of the hand, it communicated with the implant, which fired the neurons in the region of the brain corresponding to that finger. At first, the patient was able to correctly identify the location about 85 percent of the time. Then, as he got used to it, he reached 100 percent.
The mapping between brain regions and sensations and their location on the hand could be unique to each person. Or they could change day by day, requiring different calibration, but “I fully expect it to work in other people,” says Bolu Ajiboye, a Case Western Reserve University neuroengineer who was not involved in the study. He’s working on a clinical trial to restore movement, not sensation, via microelectrode stimulation of the brain.
There are several limitations to the system, Gaunt notes. For one, the test subject described some of the sensations as normal but others as electrical tingles.
Also, the team could not replicate the behavior in all parts of the hand–for example, the tips of fingers. Gaunt suggests that the implant’s electrodes may not have been positioned in quite the right spot to stimulate the needed neurons.
The research is a “proof of principle,” that “possibly natural perceptions” can appear after stimulation, says Zelma Kiss, a neurosurgeon and expert in deep-brain microstimulation at the University of Calgary in Alberta, Canada, who was also not involved.
She says it’s not clear how many microelectrode arrays are needed to cover sensation in the whole hand. The more arrays, the more implants and more wires needed.
Gaunt says his team is now working on improving performance as well as exploring how the implant can help with delicate motion tasks alongside a motion control implant.
But progress is likely to be slow. “It’s important to remember that these experiments are really, really hard,” Ajiboye says.