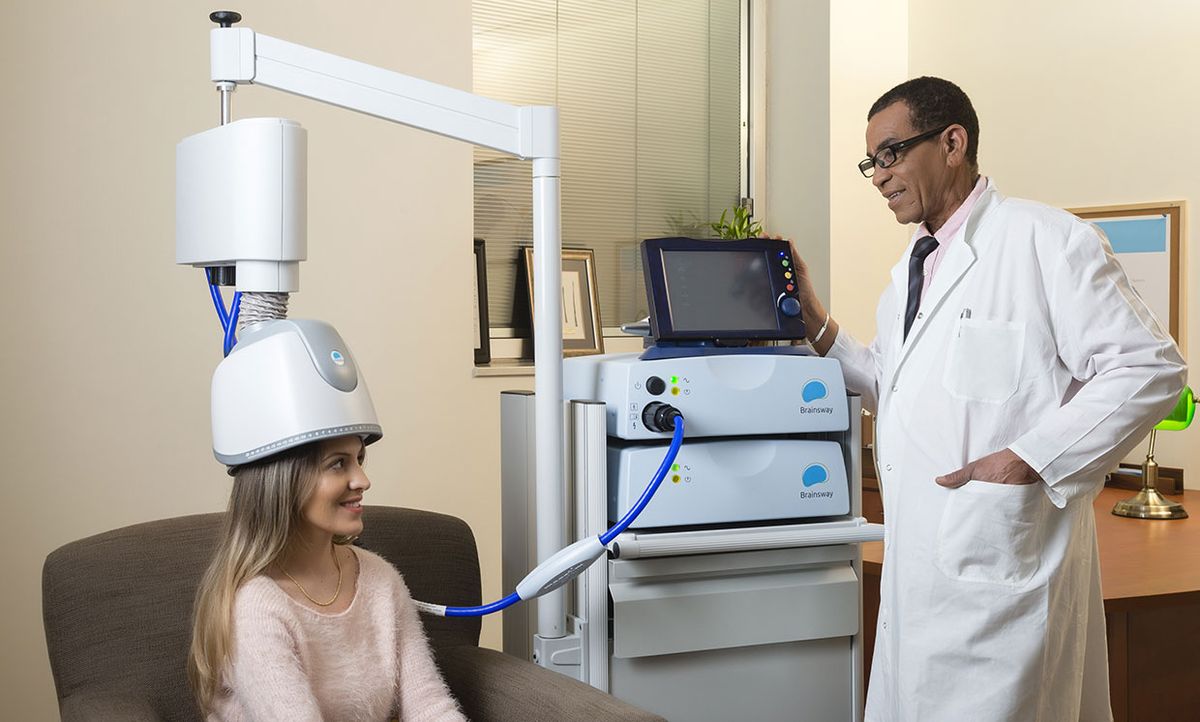
In 2013, Jerusalem-based BrainsWay began marketing a new type of brain stimulation device that uses magnetic pulses to treat major depressive disorder.
Now, thanks to positive results in a study of 100 patients, the company has received approval from the U.S. Food and Drug Administration (FDA) to market the device for a second psychiatric condition—obsessive-compulsive disorder (OCD).
According to the National Institute of Mental Health, about 1 percent of U.S. adults suffer from the anxiety disorder, characterized by uncontrollable thoughts and behaviors and the urge to repeat them over and over. For some OCD sufferers, drug and psychotherapy treatments simply don’t work.
Enter magnetic stimulation. It’s not a new idea: Various types of energy—electric fields, ultrasound, light, and magnets—have been used to stimulate the brain in the hopes of treating psychiatric conditions such as depression.
Typically, noninvasive electrical and magnetic fields applied to the scalp, such as in transcranial direct current stimulation (tDCS), affect wide swaths on the brain, activating everything in their path. The most precise methods, deep brain stimulation techniques, require implanting an electrode deep into the brain.
The new device, however, relies on deep transcranial magnetic stimulation (Deep-TMS) to stimulate deep brain structures via powerful, carefully positioned electromagnets. The BrainsWay device stimulates the brain at greater depth and breadth than other TMS devices, claims company CEO Yaacov Michlin. “This allows us to noninvasively target previously unreachable areas of the brain,” he says.

The device, which looks like a futuristic bonnet hair dryer, contains a magnetic coil that specifically targets regions in the deep front of the brain. For depression, a specialized coil targets the bilateral prefrontal cortex with preference toward the left, according to Michlin.
For OCD, they developed a new coil, called the H-7, which targets the anterior cingulate cortex, a section of the brain just under the frontal lobe that folds over the corpus callosum. Imaging studies have shown abnormalities in the frontal area of the brain in patients with OCD, but how those are linked to OCD symptoms is unclear.
The FDA’s approval was largely based on data from a clinical trial of 100 individuals with OCD at 11 medical centers. Of those, 49 patients received the BrainsWay treatment and 51 underwent a sham treatment, which included the same noises and sensation as the real device but without electromagnetic stimulation. The 25-minute treatment sessions were conducted five times per week for a total of six weeks.
By the end of the trial, 38 percent of the patients experienced a significant decrease in their OCD symptoms (by at least 30 percent) following treatment with the BrainsWay device, while only 11 percent of patients with the sham device had a similar experience. No serious side effects were noted.
The company intends to start installing the OCD helmet immediately as an upgrade to its current systems and as part of new installations, says Michlin. The device is only available for use in clinics, not home use.
According to Michlin, BrainsWay is also exploring Deep-TMS as a treatment for smoking cessation, post-traumatic stress disorder, bipolar disorder, and opioid withdrawal.
“In the longer term we hope to develop a personalized medicine approach to our treatments and have biomarkers to predict success,” he adds.
Megan is an award-winning freelance journalist based in Boston, Massachusetts, specializing in the life sciences and biotechnology. She was previously a health columnist for the Boston Globe and has contributed to Newsweek, Scientific American, and Nature, among others. She is the co-author of a college biology textbook, “Biology Now,” published by W.W. Norton. Megan received an M.S. from the Graduate Program in Science Writing at the Massachusetts Institute of Technology, a B.A. at Boston College, and worked as an educator at the Museum of Science, Boston.