Neural Electrodes Snake Around Blood Vessels, Up Nerves
Shape-memory materials could make medical devices longer lasting and easier to implant
Getting neurons to communicate with electronics has always been hard—hard on the neurons, that is. Arrays of rigid metal electrodes implanted in the brain pierce blood vessels and dislodge support cells, causing the body to cover up the array with an insulating scar, which prevents many incoming signals from getting through. Engineers now think shape-memory materials could do the job much better, because they can be programmed to snake around blood vessels and climb nerves like a vine.
One of these materials, an alloy of nickel and titanium called nitinol, easily moves between shapes depending on the temperature. A target shape can be programmed by bending cool nitinol into position, heating it to a critical temperature, and holding it at that temperature for a period of time. Thereafter, even if you bend it into another shape, heating the alloy will transform it back into the programmed shape.
Researchers in Duygu Kuzum’s lab at the University of California, San Diego, figured that electrodes made from nitinol might be a good fit for long-term brain implants. Today’s standard is an array of spikelike microelectrodes that is pressed into brain tissue, where it connects with and records signals from nearby neurons. This type has been successfully used in brain-computer interfaces that have let people control machines with their minds.
However, the implantation procedure tears at the brain’s vasculature, stresses its astrocyte support cells, and causes other damage, as UCSD graduate student Ruoyu Zhao explained to engineers in December at the IEEE Electron Devices Meeting (IEDM), in San Francisco. In response, the body walls off the area near the electrode array with scar tissue, which “leads to a dense and electrically inactive site around the implant,” he says. So the number of neurons the implant can sense drops off rapidly over time.
Kuzum and her group figured that if the microelectrodes could steer around blood vessels, the body’s response would be much less severe. The idea is to take a high-resolution CT scan of the implant area, map a safe path for the electrodes, and program microelectrodes to form the shape of that path as they’re inserted into the brain.
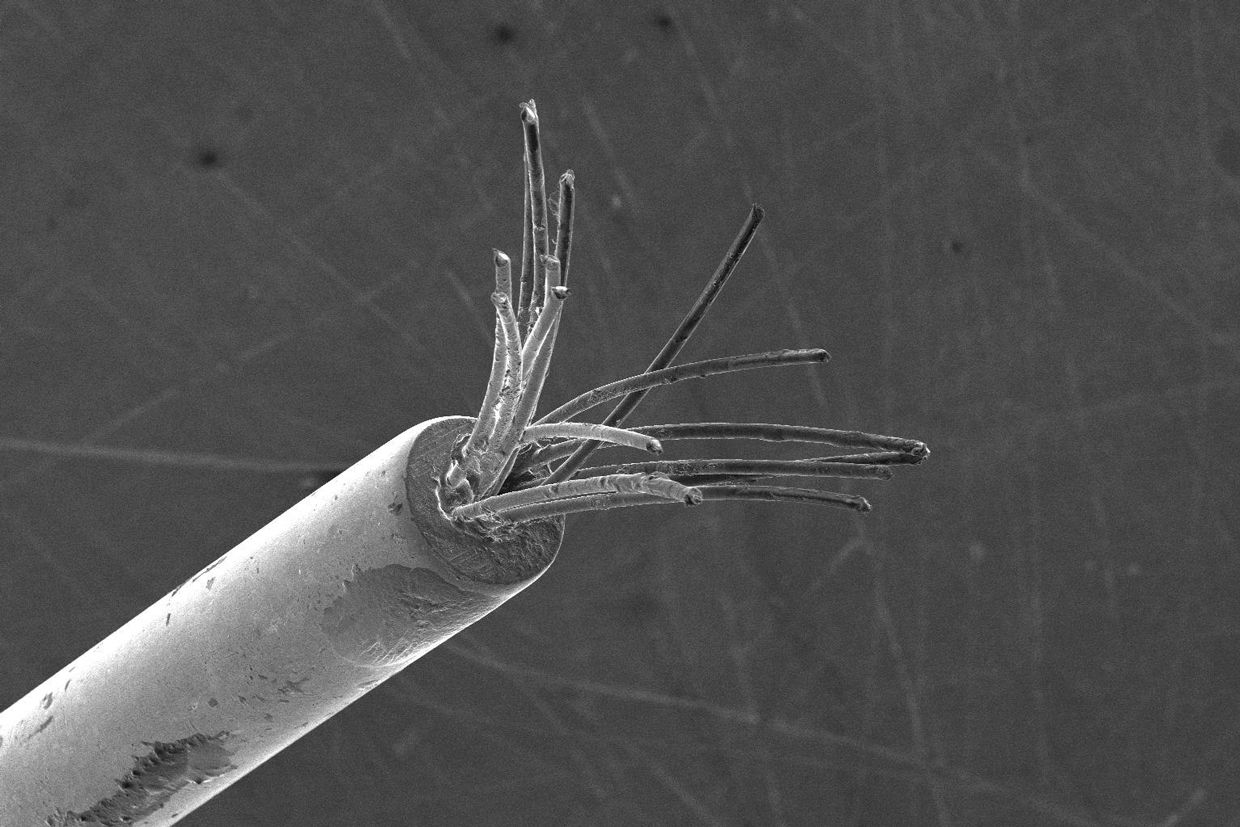
The team started by making a bundle of 16 nitinol electrodes, each with a diameter of just 23 micrometers. (The diameter of the smallest form of human neuron is about 4 μm.) The group then fitted these electrodes into a mold in the shape of the path the electrodes must travel through a model of the brain. Running 155 milliamperes through each electrode for 10 seconds heated it sufficiently to program the shape.
Kuzum’s group built an injection device that feeds a bundle of microelectrodes through a 210-μm-wide needle. They tested the system by inserting it into a 3D-printed model of a mouse brain. As the injector pushed the electrodes in, it heated them with current. This set the electrodes at their programmed shapes, bending them around the model’s blood vessels.
In a test in real mouse brains, the electrodes were able to distinguish the signals of individual neurons and caused minimal damage at the implant site. However, the UCSD team has not yet shown that the implants maintain their ability to pick up neural signals better than standard microarrays do over a long period. Kuzum says the team will do one-year implant experiments in mice in the future. Their more immediate goal is to make the electrodes even narrower, perhaps smaller than the neural cell bodies themselves.
The technology is a long way from use in humans, Kuzum says. But her team is exploring the idea with surgeons at UCSD as a possible improvement to electrodes for deep-brain stimulation, which are now implanted to treat Parkinson’s disease.
In China, a separate group of engineers—at Tsinghua University, in Beijing, and at Zhejiang University, in Hangzhou—is seeking a less-damaging, easier-to-implant electrode for nerves rather than the brain. Their project was inspired by the way vines climb poles and trees, Yingchao Zhang from Tsinghua University told engineers at IEDM. Instead of nitinol, the team used a shape-memory polymer. The substance works in a similar way, but the change in shape is triggered by body temperature.
Zhang and his colleagues constructed a 100-μm-thick substrate from a shape-memory polymer and deposited an even thinner pattern of gold onto it to conduct nerve signals. Then, they programmed the polymer to bend itself into a tight corkscrew.
In one experiment, they implanted the device on a rabbit’s vagus nerve. When the unwound polymer encountered the animal’s body heat, it snaked its way around the nerve. Zhang and his colleagues then tested the connection by stimulating the nerve to alter the rabbit’s heart rate.
Companies have explored vagus nerve stimulation to treat heart failure, though so far without commercial success. However, it is currently a treatment for epilepsy and drug-resistant depression, and is under trial for enhancing stroke recovery, and for treating Crohn’s disease, migraines, and other maladies.
Electrodes that slither through your brain and body might seem a bit creepy, but at least they will adhere to the old physician’s axiom: First, do no harm.
This article appears in the February 2019 print issue as “Shape-Shifting Electrodes for the Brain.”
Samuel K. Moore is the senior editor at IEEE Spectrum in charge of semiconductors coverage. An IEEE member, he has a bachelor's degree in biomedical engineering from Brown University and a master's degree in journalism from New York University.