Biological organisms have certain useful attributes that synthetic robots do not, such as the abilities to heal, adapt to new situations, and reproduce. Yet molding biological tissues into robots or tools has been exceptionally difficult to do: Experimental techniques, such as altering a genome to make a microbe perform a specific task, are hard to control and not scalable.
Now, a team of scientists at the University of Vermont and Tufts University in Massachusetts has used a supercomputer to design novel lifeforms with specific functions, then built those organisms out of frog cells.
The new, AI-designed biological bots crawl around a petri dish and heal themselves. Surprisingly, the biobots also spontaneously self-organize and clear their dish of small trash pellets.
“This wasn’t something that we explicitly selected for in our evolutionary algorithm,” says Josh Bongard, a roboticist at the University of Vermont who co-led the research, published this week in the Proceedings of the National Academy of Sciences. “It emerges from the fact that cells have their own intelligence and their own plans.”
The idea for AI-designed biobots came from a DARPA funding call for autonomous machines that adapt and thrive in the environment. Bongard and biologist Michael Levin at Tufts University conceived a plan to take advantage of Mother Nature’s hard work and build a machine out of something already capable of adapting: living cells.
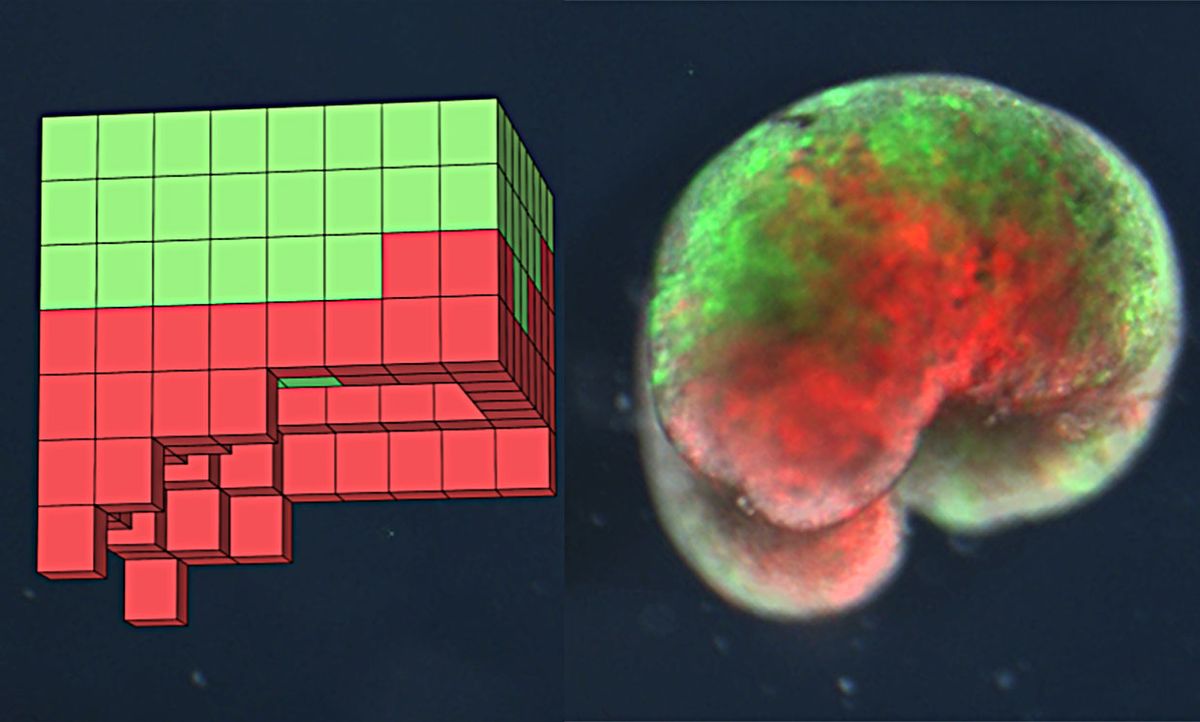
The researchers ran an evolutionary algorithm on a supercomputer at the University of Vermont over several days. The algorithm, inspired by natural selection, used biological building blocks to create a random population of new life-form candidates. The algorithm then winnowed through the designs with a fitness function that scored each candidate on its ability to do a certain thing—in this case, the ability to move.
The most promising designs became the basis to spawn a new set of designs, and the best of those were selected again. Rinse and repeat, and after 100 runs of the algorithm, tossing out billions of potential designs, the team had a set of five finalists—AI-created designs that moved well in silico.
Bongard’s team sent the finalist designs to Levin’s lab at Tufts, where microsurgeon Douglas Blackiston deemed four of the five designs too difficult or impossible to build. But the fifth design seemed doable. Blackiston used tiny forceps and a tiny electrode under a microscope to cut and join heart and skin cells from the African frog Xenopus laevis into a close approximation of the computer’s design. When cut in half, the cells stitched themselves back together—something today’s robots and computers clearly don’t do.
Once constructed, the millimeter-wide biobots moved around a petri dish as the heart cells contracted. When the team put small pellets into the dish, the cells unexpectedly worked together to clump the pellets into neat piles.
Bongard imagines a future where such biobots could be used to clean up microplastics in the ocean, especially as the biobots are 100 percent biocompatible and degrade in salt water. “That might make these biobots a uniquely appealing approach for environmental remediation,” says Bongard.
For now, the miniscule robots are best at locomotion, but Bongard has other tasks in mind. The next step, he says, is developing a “cage bot”—an empty cube to pick up and carry a payload. With that ability, one could build bots out of a person’s own cells, then use them to deliver medications deep into the body without prompting an immune response, the authors suggest.
Without a digestive system to ingest food or a nervous system to sense the surrounding environment, the organisms lived for just days. In the future, incorporating different cell types could change that: “If we wanted them to exist for longer periods of time, we might want them to be able to find and eat food sources,” says Bongard. “We’d also like to be able to incorporate sense organs into these biobots.” The collaborators are now building AI-designed biobots with mammalian cells.
The team is keenly aware that their new organisms might leave some people feeling unsettled, slipping into the uncanny valley. “Frogs that are not frogs definitely qualify for this,” says Bongard.
Plus, as they create new lifeforms—with, say, digestive, nervous, and even reproductive systems—the team is working with bioethicists and following strict animal welfare laws. “As we move further and further away from recognizable organisms, we may need to create new regulations for this kind of technology,” says Bongard.
Megan is an award-winning freelance journalist based in Boston, Massachusetts, specializing in the life sciences and biotechnology. She was previously a health columnist for the Boston Globe and has contributed to Newsweek, Scientific American, and Nature, among others. She is the co-author of a college biology textbook, “Biology Now,” published by W.W. Norton. Megan received an M.S. from the Graduate Program in Science Writing at the Massachusetts Institute of Technology, a B.A. at Boston College, and worked as an educator at the Museum of Science, Boston.