There’s been plenty of fanfare about the rise of mobile health but it’s still early days for health apps at the doctor’s office. While a third of U.S. physicians say they have recommended an app to patients, most are wary of fully integrating them into their medical practices. They’re not sure how to choose an app that really works and worry about how much time it will take busy staff to learn and coordinate app-based care.
Apple’s new CareKit, an open source software development platform set to launch in April, aims to help with at least one of these problems. CareKit will allow developers to build apps that share the same guts, some of which are intended to smooth the transfer of information between physicians and patients.
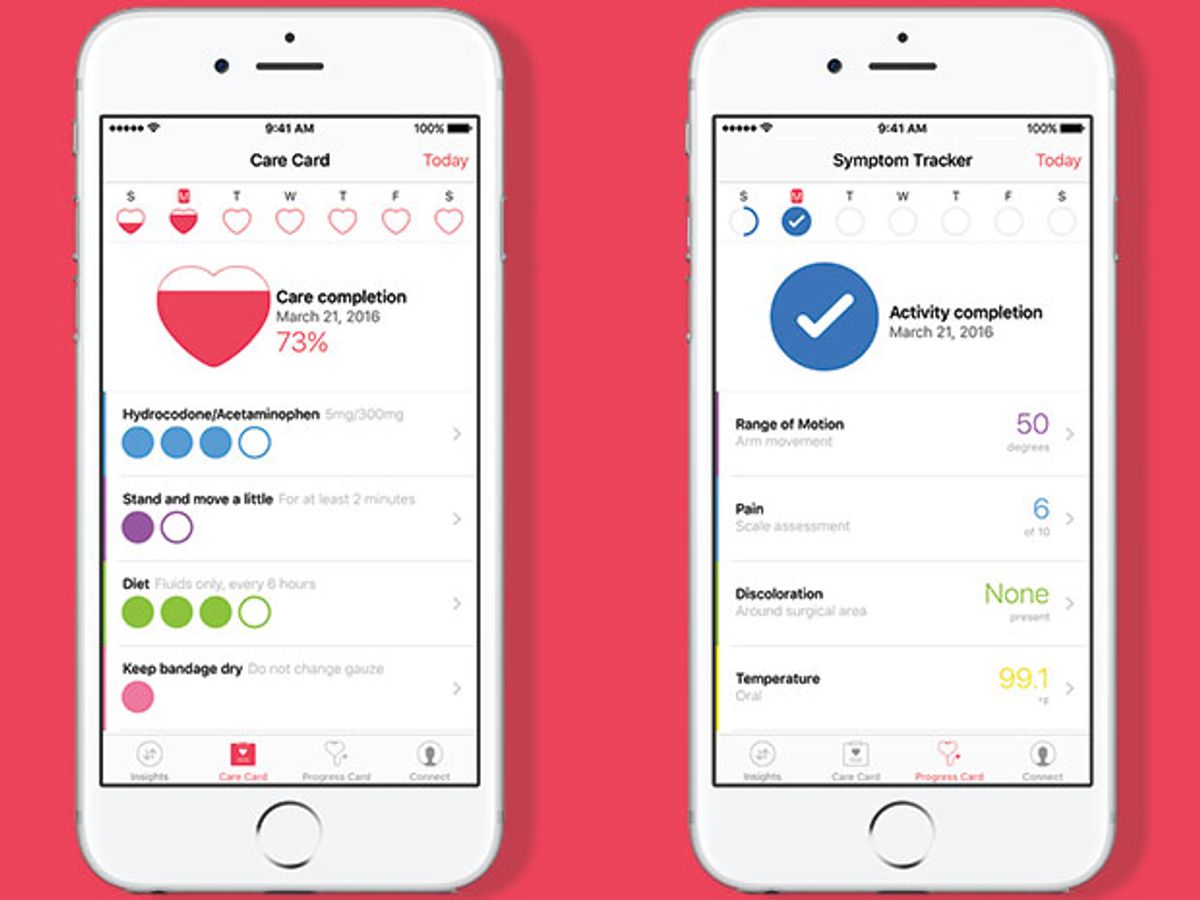
CareKit’s first four modules will include a scorecard to log whether a user has taken medicine or exercised, a symptom tracker to build surveys to record moods or side effects, a dashboard to monitor progress, and a share function for patients to send information to family, friends, and physicians.
Adam Baker, head of product and design for the health information startup Iodine, says CareKit will offer developers an easy way to build portals that physicians and patients will find familiar. “When somebody is putting treatments into our application and into other apps, it will feel the same,” he says.
Ease of use for both physicians and patients matters to developers because patients are 10 percent more likely to stick with an app for at least 30 days if their physician prescribes it to them, researchers from the IMS Institute for Healthcare Informatics have found.
Iodine, which was one of Apple’s first CareKit partners, will redevelop the company’s Start app to incorporate CareKit’s symptom tracker and share option. Start was released just five months ago and is meant to help patients monitor depression by recording which antidepressants work best for them.
However, many physicians share another concern about health apps that will persist even after the technical wrinkles of integration are ironed out–how do they know which apps will actually help their patients? There are more than 165,000 health apps available through the app stores of Google and Apple, and they represent a mix of evidence-backed programs and those that remain wholly untested.
“For all the enthusiasm, we don’t want to overhype where things are,” says Murray Aitken, executive director of the IMS Institute for Healthcare Informatics. “There are still legitimate concerns by physicians about the evidence for which apps are good.”
Most research to date has examined whether people are using apps or not, rather than taking a hard look at their effectiveness. An IMS analysis of 113 studies suggests apps might be most effective at lowering costs and improving outcomes when helping to manage diabetes, cardiovascular disease, obesity, mental health, and multiple sclerosis. Currently about two-thirds of health apps focus on fitness and wellness, while a quarter help patients monitor and track diseases.
Iodine’s Baker says preliminary data suggest that Start helps people to manage their depression, at least in the short term. Eventually, the company wants to conduct large clinical trials. “There is an effect of using the app, although we don’t fully understand what it is yet over the long term,” he adds.
Jeff Dachis, CEO and founder of a diabetes management app called One Drop that is also among Apple’s six initial CareKit partners, says the first 11 months of preliminary data fromhis company shows the app makes users more engaged in their own health, and that its medication reminders improve adherence. He also hopes to publish a peer-reviewed study at some point.
All CareKit developers will have to comply with Apple’s development guidelines for health apps, which include additional privacy protections and prohibit diagnostic apps or those that offer treatment advice unless developers have regulatory approval.
The U.S. Food and Drug Administration has approved hundreds of apps that transform a smartphone into a medical device by using the microphone as a stethoscope or sensors to take an electrocardiogram. The agency simply monitors most that it considers low risk, such as those that help people track fitness or keep a medication record.
For the moment, Baker cautions consumers to conduct due diligence and bring a healthy sense of skepticism when they enter an app store, just as they would at a doctor’s office. “Always take medication and medical information and medical apps with a grain of salt,” he says.