Researchers at the Massachusetts Institute of Technology (MIT) have developed a nanoparticle that enables both magnetic resonance imaging (MRI) as well as fluorescent imaging in living animals. The researchers believe that a single nanoparticle capable of performing these two functions should be able to help track specific molecules through the body, monitor a tumor’s environment, and determine whether drugs have reached their intended target.
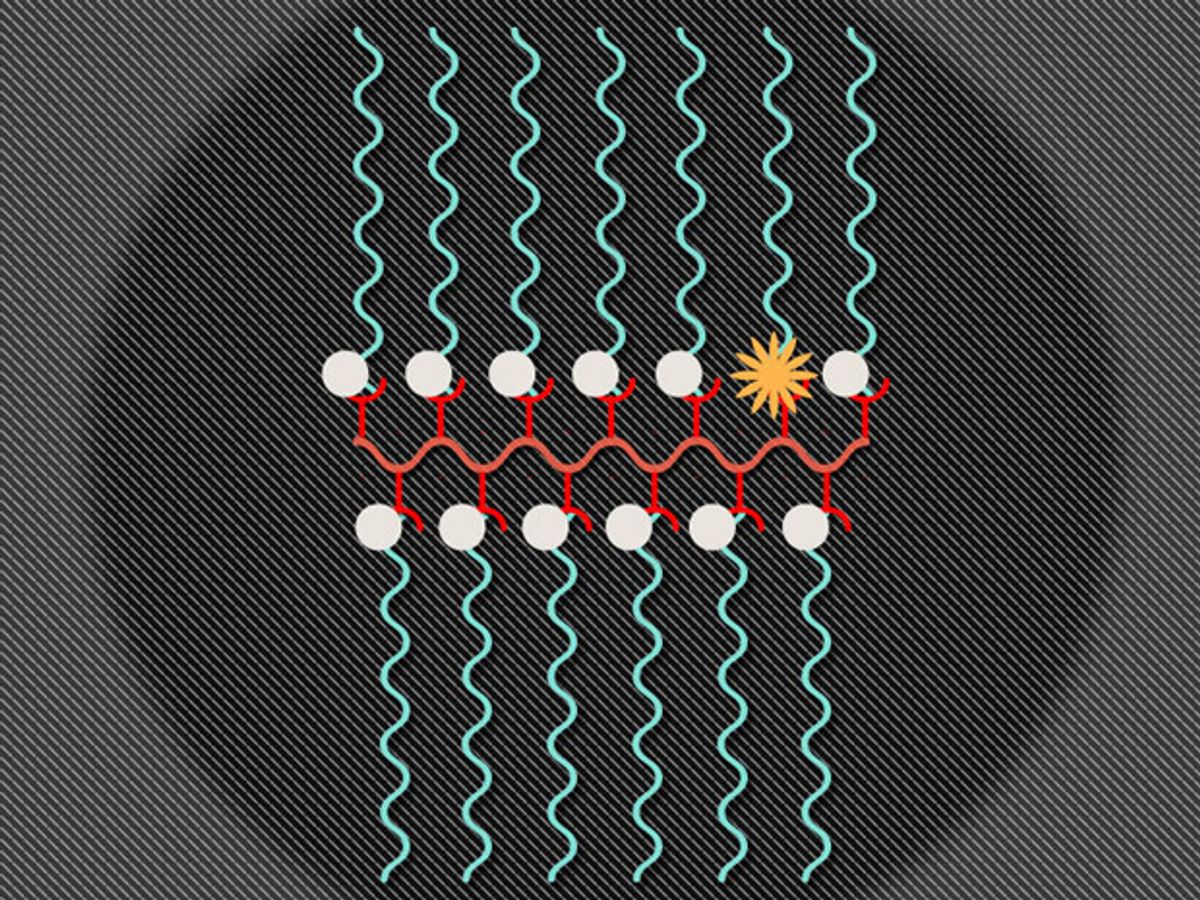
In research published in the journal Nature Communications, the MIT team combined an MRI contrasting agent called nitroxide and a fluorescent molecule called Cy5.5 to produce a nanostructure called a branched bottlebrush polymer. The ratio of the two materials in the nanoparticle is 99 percent nitroxide and 1 percent Cy5.5.
This combination enables both MRIs and fluorescent imaging because of the interesting way these materials interact with each other. The nitroxides are reactive molecules in which a nitrogen atom is bound to an oxygen atom with one unpaired electron. Typically, the nitroxides suppress the Cy5.5’s fluorescence, except when the nitroxides are in the presence of molecule from which they can grab an electron, which, in the case of this study, was a vitamin C molecule. Once the free electrons in the nitroxides bind with the free electrons from another molecule, the MRI signal switches off and the Cy5.5 fluoresces.
In the study, which used mice as subjects, the researchers found that the nanoparticles headed towards the liver, where nanoparticles typically end up. It was there that the nanoparticles came in contact with vitamin C (which is produced by the mouse's liver), the MRI signals were turned off, and the Cy5.5 began to fluoresce. Vitamin C ultimately finds its way to a mouse’s brain, and so the researchers were able to detect fluorescence there. Meanwhile, in areas where the concentration of vitamin C was low, the MRI contrast proved to be strong.
The researchers continue to work on enhancing the signal differences that occur when the nanoparticles encounter a target molecule, such as vitamin C. They have also experimented with adding up to three different drugs to the nanostructure. The aim of having these drugs hitch a ride on the nanostructure is to give diagnosticians the ability to track whether they reach their intended target. A fair amount of research is currently being done in this area, wherein nanoparticles behave as both diagnostic tools and therapeutic devices—a capability that has led to the nanoparticles being dubbed “theranostic” nanoparticles.
“That’s the advantage of our platform—we can mix and match and add almost anything we want,” said Jeremiah Johnson, an assistant professor of chemistry at MIT and senior author of the study, in a press release.
The researchers also believe that this platform should allow detection of oxygen radicals inside a patient’s tumor—a biomarker that can indicate how aggressive the tumor is.
“We think we may be able to reveal information about the tumor environment with these kinds of probes, if we can get them there,” said Johnson. “Someday you might be able to inject this in a patient and obtain real-time biochemical information about disease sites and also healthy tissues, which is not always straightforward.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.