14 November 2012—Researchers at the University of Rochester have found an efficient way to produce hydrogen fuel from water using sunlight. A light-powered hydrogen generator might replace bulky hydrogen tanks in future cars or complement photovoltaic solar cells, some scientists suggest. The system improves on previous schemes by both lasting longer and generating larger amounts of hydrogen. It might also be cheaper because it does not use a catalyst made of platinum or other precious metals, as is common in other experimental artificial photosynthesis systems.
In photosynthesis, two molecules of water are split into “reduced hydrogen equivalents” and oxygen in the presence of sunlight, storing energy. The hydrogen equivalents then enable the transformation of carbon dioxide into simple sugars. Artificial photosynthesis schemes aim to replicate the process but have focused on producing hydrogen, which is a comparatively simple process.
For hydrogen generation, a molecule is needed to absorb photons and transfer electrons to a catalyst, which in turn transfers the electrons to the water molecules’ protons, producing hydrogen. Unfortunately, in many of the artificial photosynthesis schemes that have been tried, this light-absorbing molecule decomposes quickly. Two days has been the upper limit for systems that use organic dyes or compounds containing metals like iridium as photosynthesizers. To overcome the decomposition problem, researchers looked to semiconductor nanocrystals. These are more stable, but those tested so far have produced little hydrogen.
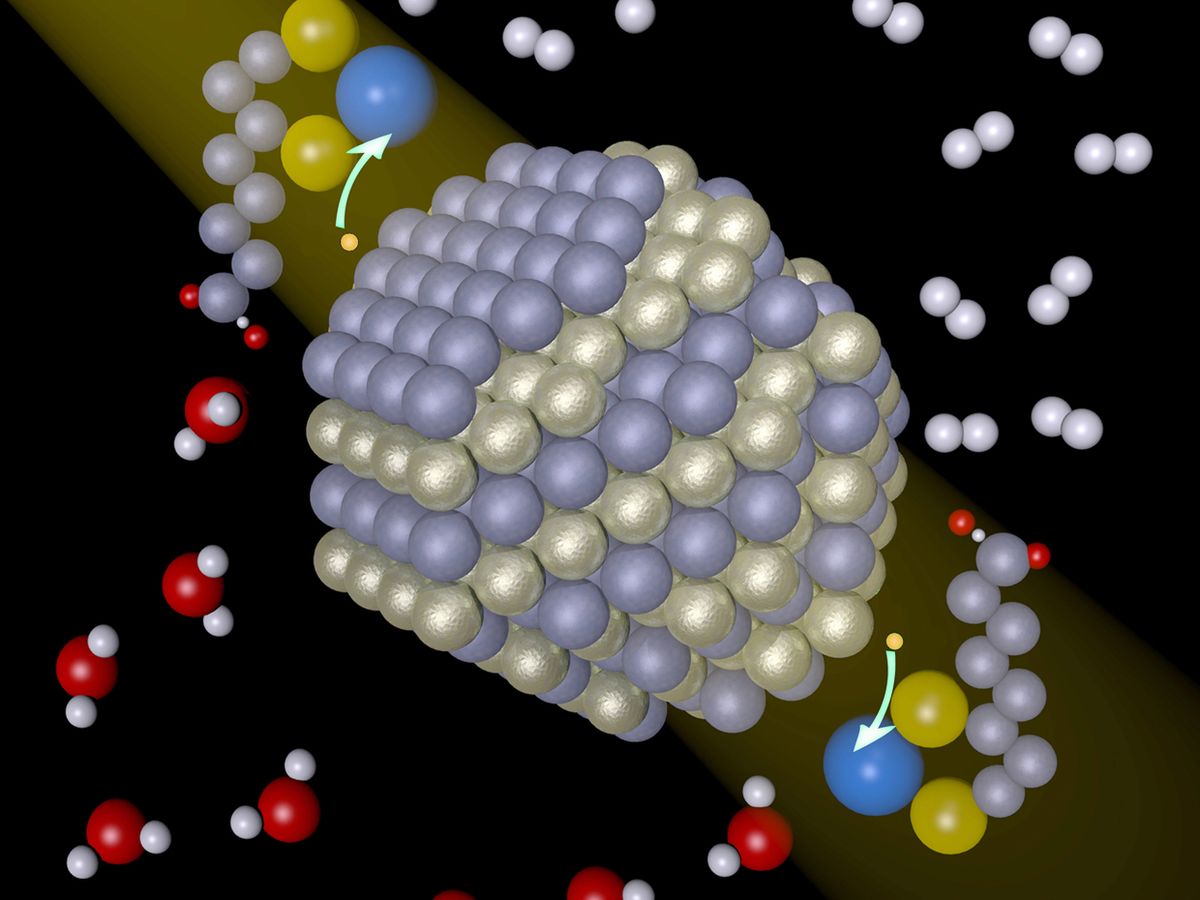
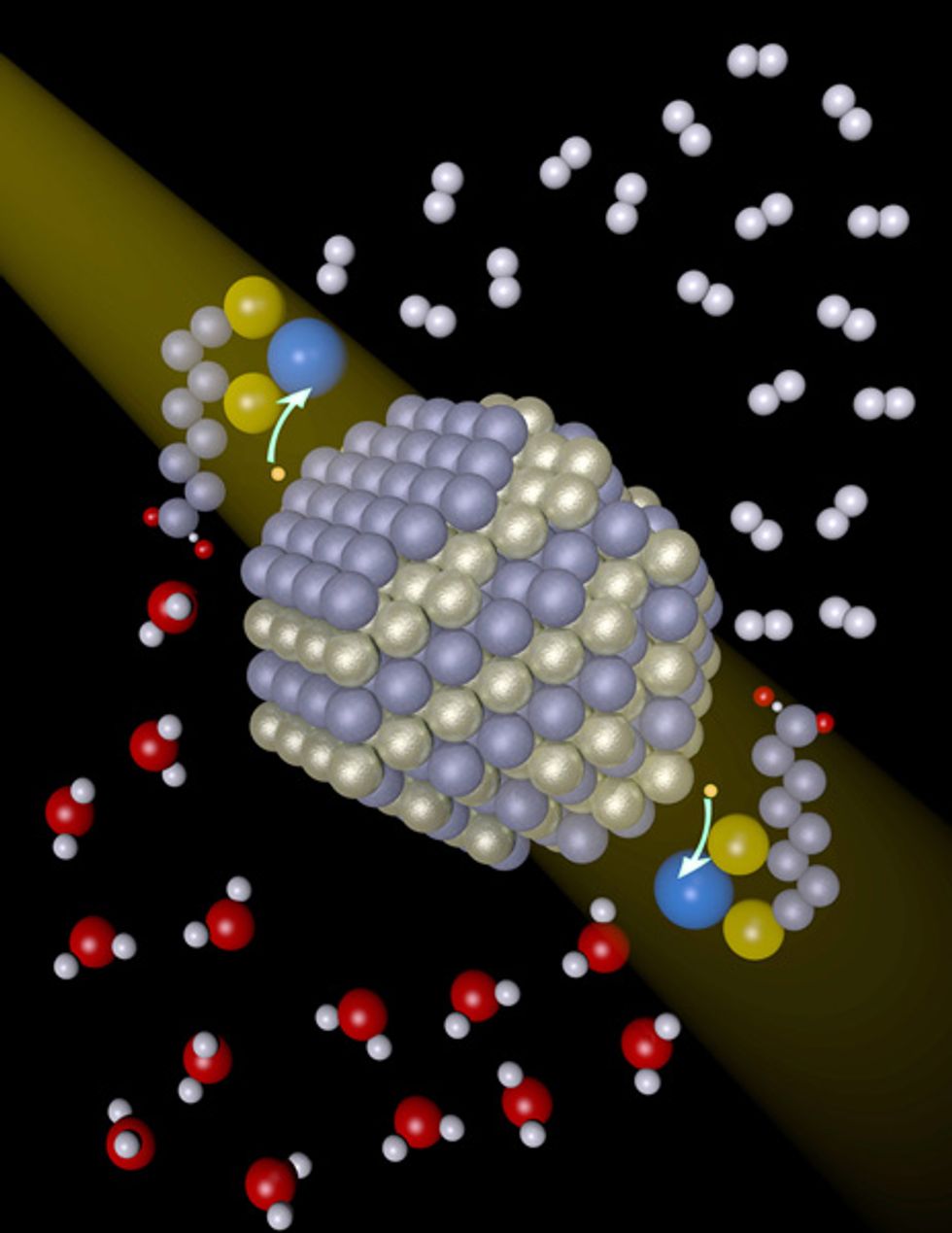
However, the new University of Rochester system, detailed last week on the website of the journal Science, had the right combination of endurance and activity. As the light absorber, the researchers used cadmium selenide particles from 2.5 to 5.5 nanometers in diameter, coated with a chemical agent to make them soluble in water. They used nickel salts as catalysts. The artificial photosynthesis system lasted for about 15 days and converted more than 7000 moles of hydrogen per mole of catalyst per hour. Another measure, quantum yield, was also high. For every 100 photons absorbed, 36 electrons were transferred to the catalyst. In natural photosynthesis, the equivalent number may be less than 10.
It is an “incredibly robust” system, says Daniel R. Gamelin, a professor of chemistry at the University of Washington, Seattle, who was not involved in the research. He also points out that the catalysts represent a shift to a less-expensive metal ion. People have combined similar nano crystals with other catalysts and generated hydrogen before, he says, but usually these were proof-of-concept studies using platinum—currently around $50 000 per kilogram—as a catalyst.
Indeed, the nickel catalysts may be the most significant tweak. In a controlled experiment, the researchers found that without nickel, the system doesn’t function. “The combination of making these nanocrystals soluble in water and having them in the presence of some nickel ion—that is the advance,” says Richard Eisenberg, who heads the group at Rochester.
The researchers don’t know all the details of how the system works. For instance, they don’t know what type of nickel ion is involved. This happens in catalysis, Gamelin says. “On the one hand, you may say it’s a shortcoming,” he says. “On the other, it’s a fantastic attribute of a catalyst that you don’t have to worry about it. You just add the ingredients, and it self-assembles.”
The next steps, according to Nathan S. Lewis at the Joint Center for Artificial Photosynthesis at Caltech, who was not involved in the Rochester research, should be “to eludicate clearly the energy return on energy invested—the thermodynamics of the process.”
Eisenberg believes hydrogen could be combined with solar cells to reduce our carbon footprint. “I’m not sure this will be the way things go, but it’s very important to have support for research on many different levels,” he says, “because something will work.”